Original Data
| Rev Diabet Stud,
2006,
3(2):76-81 |
DOI 10.1900/RDS.2006.3.76 |
The Impact of the Angiotensin-Converting Enzyme Insertion/Deletion Polymorphism on Severe Hypoglycemia in Type 2 Diabetes
Rachel M. Freathy, Kathryn F. Lonnen, Anna M. Steele, Jayne A. L. Minton, Timothy M. Frayling, Andrew T. Hattersley, Kenneth M. MacLeod
Institute of Biomedical and Clinical Science, Peninsula Medical School, Exeter, United Kingdom. The first two authors contributed equally to this work.
Address correspondence to: Kenneth MacLeod, e-mail: kenneth.macleod@pms.ac.uk
Keywords: ACE, angiotensin-converting enzyme, association, deletion, hypoglycemia, insertion, polymorphism, type 2 diabetes
Abstract
The insertion/deletion (I/D) polymorphism of the angiotensin-converting enzyme gene (ACE) is associated with altered serum ACE activity. Raised ACE levels and the ACE DD genotype are associated with a 3.2 to 6.8-fold increased risk of severe hypoglycemia in type 1 diabetes. This relationship has not been assessed in type 2 diabetes. We aimed to test for association of the ACE I/D polymorphism with severe hypoglycemia in type 2 diabetes. Patients with type 2 diabetes (n = 308), treated with insulin (n = 124) or sulphonylureas (n = 184), were classified according to whether or not they had previously experienced severe hypoglycemia. Samples of DNA were genotyped for the ACE I/D polymorphism using two alternative polymerase chain reactions to prevent mistyping due to preferential amplification of the D allele. Overall, 12% of patients had previously experienced one or more episodes of severe hypoglycemia. This proportion did not differ between genotype groups (odds ratio (95% confidence limits) for carriers of D allele relative to II homozygotes: 0.79 (0.35-1.78)). This study found no evidence for association of the ACE I/D polymorphism with severe hypoglycemia frequency in patients with type 2 diabetes. However, we cannot rule out a smaller effect (odds ratio ≤ 1.78). Our results suggest that any effect of ACE genotype on severe hypoglycemia risk in type 2 patients is likely to be smaller than that seen in type 1 diabetes. We recommend future larger-scale studies.
Introduction
Hypoglycemia is the limiting factor in the achievement of optimal glycemic control for patients with diabetes [1] and can cause unpleasant symptoms, fear, anxiety, abandonment of treatment plans or increase the risk of accident and injury [2]. Less attention has been given to hypoglycemia in patients with type 2 diabetes than type 1 diabetes [2]. This is probably due to the lower frequency of, and mortality from, hypoglycemic events in patients with type 2 diabetes [1]. Estimates of the frequency of severe hypoglycemia (requiring assistance from another person to effect recovery) in type 2 diabetes vary greatly, from 2 to 73 episodes per 100 patient years [3-6] compared with 62 to 180 episodes per 100 patient years in type 1 diabetes [3, 7-9]. Hypoglycemia incidence in type 2 diabetes is higher in patients treated with insulin [10] and there is evidence that the frequency of severe hypoglycemia is similar in patients with type 1 and type 2 diabetes matched for duration of insulin therapy [11]. In view of the above, and since 90% of all people with diabetes have type 2 diabetes [2], the study of hypoglycemia in this group arguably deserves as much attention as in type 1 diabetes.
A common polymorphism in the gene for angiotensin-converting enzyme (ACE), located on chromosome 17q23.3, is reproducibly associated with severe hypoglycemia in type 1 diabetes [8, 12]. Situated in intron 16, the polymorphism is characterized by the insertion (I) or deletion (D) of a 287-bp Alu repeat element [13, 14]. The I and D alleles occur with roughly equal frequency in the U.K. population [15]. The D allele results in higher serum ACE concentrations than the I allele [8, 12, 13, 16], accounting for 47% of the total phenotypic variance in serum ACE [13]. ACE is expressed in a wide range of tissues and the enzyme is present in the circulation [17]. As part of the renin-angiotensin system, ACE is responsible for the conversion of angiotensin I to angiotensin II (AII) [17].
Multiple associations have been reported with the ACE I/D polymorphism. The D allele is associated both with increased serum ACE levels and risk of severe hypoglycemia (retrospectively and prospectively) in type 1 diabetes [8, 12]. In addition, serum ACE and severe hypoglycemia in type 1 diabetes are significantly correlated [8, 9, 12]. The polymorphism has been studied in relation to type 2 diabetes and correlated with the presence of macrovascular [18, 19] and microvascular complications [20-22] and with the progression of diabetic nephropathy [23, 24]. However, much of this evidence remains controversial [17, 18, 25, 26]. The ACE I/D polymorphism may be associated with insulin sensitivity. Patients with type 2 diabetes or healthy or overweight subjects carrying the D allele or DD genotype have increased insulin sensitivity [27-30]. However, these studies were small (n ≈ 100) and the association is not always seen [31].
The ACE I/D polymorphism has not been studied in relation to severe hypoglycemia in type 2 diabetes. We aimed to carry out a preliminary assessment of the relationship between ACE genotype and frequency of severe hypoglycemia in patients with type 2 diabetes.
Methods
Subjects, clinical characteristics and hypoglycemia questionnaire
Patients were recruited from Exeter (U.K.) and the surrounding region as part of a community-based collection of patients with diabetes. The inclusion criteria for this particular study were that patients should have type 2 diabetes (age at diagnosis ≥ 25 years; diagnosed by consultant diabetologist) and be treated for this with either insulin or sulphonylurea medication. Even though hypoglycemia may differ clinically between patients on insulin and sulphonylureas, we included both groups because there are no previous studies on the effect of the ACE I/D polymorphism on severe hypoglycemia in type 2 diabetes and therefore no evidence to suggest that an effect of genotype would differ between the two groups. Our patients were recruited from outpatients clinics and general practice centres. Given that a large proportion of these patients take ACE inhibitor or AII receptor blocker medication, this group was not excluded. The clinical characteristics of subjects are shown in Table 1. This work conforms to the ethical guidelines of the World Medical Association Declaration of Helsinki. It was approved by the North and East Devon Local Research Ethics Committee and all patients gave their informed consent.
Table
1.
Clinical characteristics of patients |
|
 |
 |
Legend:
Continuous data are given as median (interquartile range). ACE: angiotensin-converting enzyme. AII: angiotensin II. * BMI data available for 279 patients. |
|
Information regarding treatment, hypoglycemia frequency and complications was collected from patients using a written questionnaire or interview. Definitions of mild (self-treated) and severe (requiring help from another person to effect recovery) hypoglycemia were given at the top of the questionnaire or explained during the interview. Enquiry was made about the frequency of severe hypoglycemia using the following categories: never; < 1 per year; 1-3 episodes per year; 4-12 per year; > 1 per month; > 1 per week. However, only 37 individuals (12%) had ever previously experienced severe hypoglycemia (9 II; 18 ID; 10 DD). Of these, 21 patients reported 1-2 episodes in total, 4 reported 3-4 episodes, 5 patients reported 5 or more episodes and 7 did not specify. Owing to this small affected percentage, data were simply analyzed according to whether patients had or had not ever previously experienced an episode. These data were collected without prior knowledge of a patient’s genotype.
Genotyping and quality control
Genomic DNA was isolated from leukocytes using standard techniques. The ACE genotype of each subject was determined by two polymerase chain reactions (PCRs) and was performed without knowledge of patients’ clinical characteristics. In the first PCR, a pair of primers was used to amplify the region of intron 16 which contains the 287-bp insertion/deletion polymorphism [13]. These were 5'-CTG GAG ACC ACT CCC ATC CTT TCT-3' (forward) and 5'-GAT GTG GCC ATC ACA TTC GTC AGA T-3' (reverse). The first PCR was performed in a final volume of 25 μl containing 50 ng genomic DNA, 20 pmol each primer, 2 mmol/l MgCl2, 0.25 mmol/l each of deoxy-ATP, -CTP, -GTP and -TTP and 0.5 U Amplitaq Gold DNA polymerase (Applied Biosystems, Warrington, UK). The reaction started with 15 min denaturation at 95˚C, followed by 35 cycles of denaturation at 94˚C for 1 min, annealing at 58˚C for 1 min and extension at 72˚C for 1 min. This was completed by a final extension at 72˚C for 10 min. The PCR products were visualized with ethidium bromide staining after electrophoresis on a 2% agarose gel. Products containing the insertion allele (I) were visible as a band at approximately 490 bp, whilst deletion (D) products were visible at approximately 190 bp.
Due to preferential amplification of the D allele, it is possible that ID heterozygotes may be mistyped as DD homozygotes [32]. Thus the second PCR was an I-specific amplification method, which was used on all samples initially typed as DD. This produced a 335 bp amplicon only in the presence of the I allele. The primers were 5'-TGG GAC CAC AGC GCC CGC CAC TAC-3' (forward) and 5'-TCG CCA GCC CTC CCA TGC CCA TAA-3' (reverse). PCR reaction conditions were as described above, apart from the annealing temperature, which was increased to 67˚C. Products were visualized with ethidium bromide staining after electrophoresis on a 2% agarose gel. All genotypes were scored independently by two individuals, and ambiguous genotypes were repeated. 75 randomly selected samples (21%) were retyped to check for inconsistencies. Inconsistencies in results were only seen in genotyping of DD subjects with the I-specific PCR, where the absence of product from an individual is consistent with either the absence of the I allele or a failure of amplification. As a result all samples scored as DD in the first PCR were all genotyped using the I-specific PCR at least twice.
Statistical analysis
We used Chi-square tests to test for association between ACE genotype and severe hypoglycemia and various clinical characteristics, and to calculate odds ratios and P values. We also used Chi-square tests to compare treatment groups with regard to prior experience of severe hypoglycemia. Age of patient, age at diagnosis and BMI were compared among genotype groups using univariate ANOVA (all genotypes) and the independent samples t test (DD/ID versus II). BMI data were log-transformed to normalize. Data on diabetes duration were compared among genotype groups using the Kruskal-Wallis test (all genotypes) and the Mann-Whitney U test (DD/ID versus II).
Our sample of 308 patients gave us 80% power to detect odds ratios (ORs) for alleles of 2.05 at p < 0.05 (two-tailed), based on a D allele frequency of 0.53 [15]. For comparison with the results of Pedersen-Bjergaard et al. [8] we had 70% power to detect ORs for the DD vs II genotype groups of 3.2 at p < 0.05 (one-tailed). These power calculations assume that 12% of subjects had previously experienced severe hypoglycemia.
Results
Genotyping and quality control
The overall genotyping assay success rate was 99%. Genotypes were in Hardy-Weinberg Equilibrium (Chi-square p = 0.16). Frequencies of the I (0.48) and D (0.52) alleles were similar to those observed in previous studies [8, 15].
ACE genotype and severe hypoglycemia
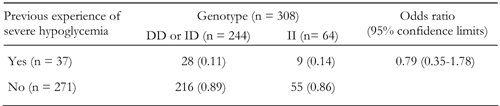
There was no association of the presence of the D allele with previous severe hypoglycemia experience (OR: 0.79 (0.35-1.78); p = 0.32 (2-tailed); Table 2). DD homozygotes had no more risk of severe hypoglycemia than II homozygotes (OR: 0.90 (0.34-2.37); p = 0.42 (1-tailed).
Table
2.
Result of test for association of the presence of the D allele with severe hypoglycemia |
|
 |
 |
Legend:
Numbers (and frequencies) of patients with or without previous severe hypoglycemia experience are divided according to genotype group. The odds ratio is calculated for presence versus absence of the D allele. |
|
Patients treated with insulin had a higher frequency of previous severe hypoglycemia (23 insulin-treated patients had experienced one or more episodes, compared with 14 sulphonylurea-treated; OR: 2.77 (1.36-5.62); p = 0.004 (2-tailed)). However, there remained no association between genotype and severe hypoglycemia experience when patients were stratified according to treatment for diabetes (insulin (OR for DD/ID versus II: 0.66 (0.23-1.89)); sulphonylurea (OR: 0.99 (0.26-3.72))). Similarly, there was no association between genotype and severe hypoglycemia experience when patients were stratified according to treatment targeted at ACE or the AII receptor (ACE inhibitor or AII receptor blocker medication (OR for DD/ID versus II: 0.57 (0.20-1.61)); no medication (OR: 1.20 (0.32-4.52))).
There was no association of genotype with sex, age, duration of diabetes, age at diagnosis, BMI, ACE inhibitor/AII receptor blocker medication or diabetes medication (p > 0.05).
Discussion
We have found no evidence that the ACE gene insertion/deletion polymorphism is associated with risk of severe hypoglycemia in type 2 diabetes. This is contrary to studies in type 1 diabetes, which have shown relative risks of 3.2 for DD relative to II homozygotes [8], or 5.3 and 6.8 for the ID and DD genotypes respectively in C-peptide-negative patients [12]. Our results suggest that we can exclude large effects (OR > 1.78) of the presence of the D allele on severe hypoglycemia risk in type 2 diabetes.
There are potential weaknesses in this study. Firstly, although our study was larger than similar association studies in type 1 diabetes [8, 12], it was relatively underpowered due to the lower frequency of severe hypoglycemia in type 2 diabetes. Since estimates of severe hypoglycemia frequency in type 2 diabetes vary widely [3-6], it was difficult to predict a priori the necessary sample size. Using the figure for severe hypoglycemia frequency obtained from the present study, we estimate that in future studies, 3000 patients with type 2 diabetes would be required for 80% power to detect ORs for alleles of 1.25 at p < 0.05 (2-tailed; D allele frequency = 0.53). It may be possible to reduce this by selecting type 2 patients at higher risk of hypoglycemia: over time, and with diminishing endogenous insulin production and glucose counter-regulation, the risk in type 2 diabetes approaches that of type 1 [1, 10, 33]. The strongest effect of ACE genotype or serum ACE activity on severe hypoglycemia risk in type 1 diabetes occurs in C-peptide-negative subjects who are already at higher risk of severe hypoglycemia [8, 12]. We found no evidence that the ACE I/D polymorphism is associated with severe hypoglycemia in our insulin-treated patients, whom we confirmed were at higher risk of severe hypoglycemia than those on sulphonylurea medication. A second caveat, again due to the lower frequency of severe hypoglycemia in type 2 diabetes, is that we were unable to use data on the number of episodes reported per person. Pedersen-Bjergaard et al. [8] found that the differences between genotype groups were primarily due to a higher number of episodes per patient with the DD genotype reporting severe hypoglycemia, whereas the proportion of people reporting at least one episode did not differ significantly between genotype groups in their study. A third potential limitation is that over half of the patients in our sample were on ACE inhibitor or AII receptor blocker medication, which may modulate ACE activity. Since the effect of ACE genotype on severe hypoglycemia risk in type 1 diabetes is believed to act through its effect on serum ACE concentrations [8, 12], any effect of genotype may have been masked in these patients [8]. However, in the original study by Pedersen-Bjergaard et al. only 55 patients on such medication were genotyped, and although no association was seen between severe hypoglycemia risk and genotype in this group, there was not sufficient statistical power for their 95% confidence intervals to exclude the effect seen in the non-treated individuals [8]. The original study also showed no difference in severe hypoglycemia risk between those treated with ACE inhibitor/ARB medication and the non-treated group (RR 0.85 (0.5-1.6; p = 0.6), even though serum ACE activity was significantly lower in the former group (p < 0.0001). As our study is the first to explore the association between ACE genotype and type 2 diabetes, we did not feel there was sufficient evidence to exclude the large proportion of patients on ACE inhibitors or ARBs, but recognized that it was imperative to do a stratified analysis. We found no evidence that the ACE I/D polymorphism is associated with severe hypoglycemia in patients not treated with ACE inhibitor or AII receptor blocker medication.
To conclude, this is the first study to examine the potential association of the ACE gene insertion/deletion polymorphism with severe hypoglycemia in type 2 diabetes. With ample power to detect the odds ratio seen in type 1 diabetes [8], we have found no evidence for an association, but we do not have the power to exclude a small effect.
Acknowledgments:
R. M. Freathy holds a Diabetes U. K. research studentship. A. T. Hattersley is a Wellcome Trust Research Leave Fellow.
References
- Cryer PE. Hypoglycaemia: the limiting factor in the glycaemic management of type I and type II diabetes. Diabetologia 2002. 45:937-948. [DOD] [CrossRef]
- Murata GH, Duckworth WC, Hoffman RM, Wendel CS, Mohler MJ, Shah JH. Hypoglycemia in type 2 diabetes: a critical review. Biomed Pharmacother 2004. 58:551-559. [DOD] [CrossRef]
- MacLeod KM, Hepburn DA, Frier BM. Frequency and morbidity of severe hypoglycaemia in insulin-treated diabetic patients. Diabet Med 1993. 10:238-245. [DOD]
- Abraira C, Colwell JA, Nuttall FQ, Sawin CT, Nagel NJ, Comstock JP, Emanuele NV, Levin SR, Henderson W, Lee HS. Veterans Affairs Cooperative Study on glycemic control and complications in type II diabetes (VA CSDM). Results of the feasibility trial. Veterans Affairs Cooperative Study in Type II Diabetes. Diabetes Care 1995. 18:1113-1123. [DOD]
- Gurlek A, Erbas T, Gedik O. Frequency of severe hypoglycaemia in type 1 and type 2 diabetes during conventional insulin therapy. Exp Clin Endocrinol Diabetes 1999. 107:220-224. [DOD]
- Murata GH, Duckworth WC, Shah JH, Wendel CS, Mohler MJ, Hoffman RM. Hypoglycemia in stable, insulin-treated veterans with type 2 diabetes: a prospective study of 1662 episodes. J Diabetes Complications 2005. 19:10-17. [DOD]
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993. 329:977-986. [DOD] [CrossRef]
- Pedersen-Bjergaard U, Agerholm-Larsen B, Pramming S, Hougaard P, Thorsteinsson B. Activity of angiotensin-converting enzyme and risk of severe hypoglycaemia in type 1 diabetes mellitus. Lancet 2001. 357:1248-1253. [DOD] [CrossRef]
- Nordfeldt S, Samuelsson U. Serum ACE predicts severe hypoglycemia in children and adolescents with type 1 diabetes. Diabetes Care 2003. 26:274-278. [DOD]
- U. K. Prospective Diabetes Study Group. U.K. prospective diabetes study 16. Overview of 6 years' therapy of type II diabetes: a progressive disease. Diabetes 1995. 44:1249-1258. [DOD]
- Hepburn DA, MacLeod KM, Pell AC, Scougal IJ, Frier BM. Frequency and symptoms of hypoglycaemia experienced by patients with type 2 diabetes treated with insulin. Diabet Med 1993. 10:231-237. [DOD]
- Pedersen-Bjergaard U, Agerholm-Larsen B, Pramming S, Hougaard P, Thorsteinsson B. Prediction of severe hypoglycaemia by angiotensin-converting enzyme activity and genotype in type 1 diabetes. Diabetologia 2003. 46:89-96. [DOD]
- Rigat B, Hubert C, Alhenc-Gelas F, Cambien F, Corvol P, Soubrier F. An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. J Clin Invest 1990. 86:1343-1346. [DOD]
- Rieder MJ, Taylor SL, Clark AG, Nickerson DA. Sequence variation in the human angiotensin converting enzyme. Nat Genet 1999. 22:59-62. [DOD] [CrossRef]
- Keavney B, McKenzie C, Parish S, Palmer A, Clark S, Youngman L, Delepine M, Lathrop M, Peto R, Collins R. Large-scale test of hypothesised associations between the angiotensin-converting-enzyme insertion/deletion polymorphism and myocardial infarction in about 5000 cases and 6000 controls. International Studies of Infarct Survival (ISIS) Collaborators. Lancet 2000. 355:434-442. [DOD]
- McKenzie CA, Abecasis GR, Keavney B, Forrester T, Ratcliffe PJ, Julier C, Connell JM, Bennett F, McFarlane-Anderson N, Lathrop GM, et al. Trans-ethnic fine mapping of a quantitative trait locus for circulating angiotensin I-converting enzyme (ACE). Hum Mol Genet 2001. 10:1077-1084. [DOD] [CrossRef]
- Kennon B, Petrie JR, Small M, Connell JM. Angiotensin-converting enzyme gene and diabetes mellitus. Diabet Med 1999. 16:448-458. [DOD] [CrossRef]
- Fujisawa T, Ikegami H, Shen GQ, Yamato E, Takekawa K, Nakagawa Y, Hamada Y, Ueda H, Rakugi H, Higaki J. Angiotensin I-converting enzyme gene polymorphism is associated with myocardial infarction, but not with retinopathy or nephropathy, in NIDDM. Diabetes Care 1995. 18:983-985. [DOD]
- Wong TY, Szeto CC, Chow KM, Chan JC, Li PK. Contribution of gene polymorphisms in the renin-angiotensin system to macroangiopathy in patients with diabetic nephropathy. Am J Kidney Dis 2001. 38:9-17. [DOD]
- Doi Y, Yoshizumi H, Yoshinari M, Iino K, Yamamoto M, Ichikawa K, Iwase M, Fujishima M. Association between a polymorphism in the angiotensin-converting enzyme gene and microvascular complications in Japanese patients with NIDDM. Diabetologia 1996. 39:97-102. [DOD]
- Ohno T, Kawazu S, Tomono S. Association analyses of the polymorphisms of angiotensin-converting enzyme and angiotensinogen genes with diabetic nephropathy in Japanese non-insulin-dependent diabetics. Metabolism 1996. 45:218-222. [DOD] [CrossRef]
- Jeffers BW, Estacio RO, Raynolds MV, Schrier RW. Angiotensin-converting enzyme gene polymorphism in non-insulin dependent diabetes mellitus and its relationship with diabetic nephropathy. Kidney Int 1997. 52:473-477. [DOD]
- Fava S, Azzopardi J, Ellard S, Hattersley AT. ACE gene polymorphism as a prognostic indicator in patients with type 2 diabetes and established renal disease. Diabetes Care 2001. 24:2115-2120. [DOD]
- Solini A, Dalla Vestra M, Saller A, Nosadini R, Crepaldi G, Fioretto P. The angiotensin-converting enzyme DD genotype is associated with glomerulopathy lesions in type 2 diabetes. Diabetes 2002. 51:251-255. [DOD]
- Nakajima S, Baba T, Yajima Y. Is ACE gene polymorphism a useful marker for diabetic albuminuria in Japanese NIDDM patients? Diabetes Care 1996. 19:1420-1422. [DOD]
- Ringel J, Beige J, Kunz R, Distler A, Sharma AM. Genetic variants of the renin-angiotensin system, diabetic nephropathy and hypertension. Diabetologia 1997. 40:193-199. [DOD] [CrossRef]
- Katsuya T, Horiuchi M, Chen YD, Koike G, Pratt RE, Dzau VJ, Reaven GM. Relations between deletion polymorphism of the angiotensin-converting enzyme gene and insulin resistance, glucose intolerance, hyperinsulinemia, and dyslipidemia. Arterioscler Thromb Vasc Biol 1995. 15:779-782. [DOD]
- Panahloo A, Andres C, Mohamed-Ali V, Gould MM, Talmud P, Humphries SE, Yudkin JS. The insertion allele of the ACE gene I/D polymorphism. A candidate gene for insulin resistance? Circulation 1995. 92:3390-3393. [DOD]
- Paolisso G, Tagliamonte MR, De Lucia D, Palmieri F, Manzella D, Rinaldi C, Bossone A, Colaizzo D, Margaglione M, Varricchio M. ACE gene polymorphism and insulin action in older subjects and healthy centenarians. J Am Geriatr Soc 2001. 49:610-614. [DOD] [CrossRef]
- Ryan AS, Nicklas BJ, Berman DM, Ferrell RE. The insertion/deletion polymorphism of the ACE gene is related to insulin sensitivity in overweight women. Diabetes Care 2001. 24:1646-1652. [DOD]
- Jeng JR, Shieh SM, Harn HJ, Lee MM, Sheu WH, Jeng CY. Angiotensin I converting enzyme gene polymorphism and insulin resistance in patients with hypertension. J Hypertens 1997. 15:963-968. [DOD] [CrossRef]
- Shanmugam V, Sell KW, Saha BK. Mistyping ACE heterozygotes. PCR Methods Appl 1993. 3:120-121. [DOD]
- United Kingdom Prospective Diabetes Study Group. United Kingdom Prospective Diabetes Study 24: a 6-year, randomized, controlled trial comparing sulfonylurea, insulin, and metformin therapy in patients with newly diagnosed type 2 diabetes that could not be controlled with diet therapy. Ann Intern Med 1998. 128:165-175. [DOD]
This article has been cited by other articles:

|
Hypoglycaemia: current management and controversies
Choudhary P, Amiel SA
Postgrad Med J 2011. 87:298-306
|
|

|
Angiotensin-converting enzyme insertion/deletion polymorphism and severe hypoglycemia complicating type 2 diabetes: the Fremantle Diabetes Study
Davis WA, Brown SG, Jacobs IG, Bulsara M, Beilby J, Bruce DG, Davis TM
J Clin Endocrinol Metab 2011. Feb 2, epub ahead of print
|
|

|
Hypoglycemia, S-ACE and ACE genotypes in a Danish nationwide population of children and adolescents with type 1 diabetes
Johannesen J, Svensson J, Bergholdt R, Eising S, Gramstrup H, Frandsen E, Dick-Nielsen J, Hansen L, Pociot F, Mortensen HB
Pediatr Diabetes 2011. 12(2):100-106
|
|

|
Angiotensin-converting enzyme and angiotensin II receptor subtype 2 genotypes in type 1 diabetes and severe hypoglycaemia requiring emergency treatment: a case cohort study
Pedersen-Bjergaard U, Nielsen SL, Akram K, Perrild H, Nordestgaard BG, Montgomery HE, Pramming S, Thorsteinsson B
Pharmacogenet Genomics 2009. 19(11):865-868
|
|

|
Serum angiotensin-converting enzyme and frequency of severe hypoglycaemia in Type 1 diabetes: does a relationship exist?
Zammitt NN, Geddes J, Warren RE, Marioni R, Ashby JP, Frier BM
Diabet Med 2007. 24 (12):1449-1454
|
|
|